|
|
등록일:
2004-04-14 10:51:44 , 조회:
3,080 |
(임상1) 건강한 성인 남자에게서의 단회 및 반복 투여 시험1)
대상: 아토피피부염 환자가 아닌 일반인. 22-42세. 14명
시행약물: 프로토픽(타크로리무스) 0.03%, 0.1%, 0.3%(임상용), 기제
방법: 1일 1회 4일간 1000cm2 도포
결과: 혈중에서 타크로리무스의 농도가 측정되지 않음.
프로토픽은 건강한 피부에서는 흡수가 되지 않습니다.
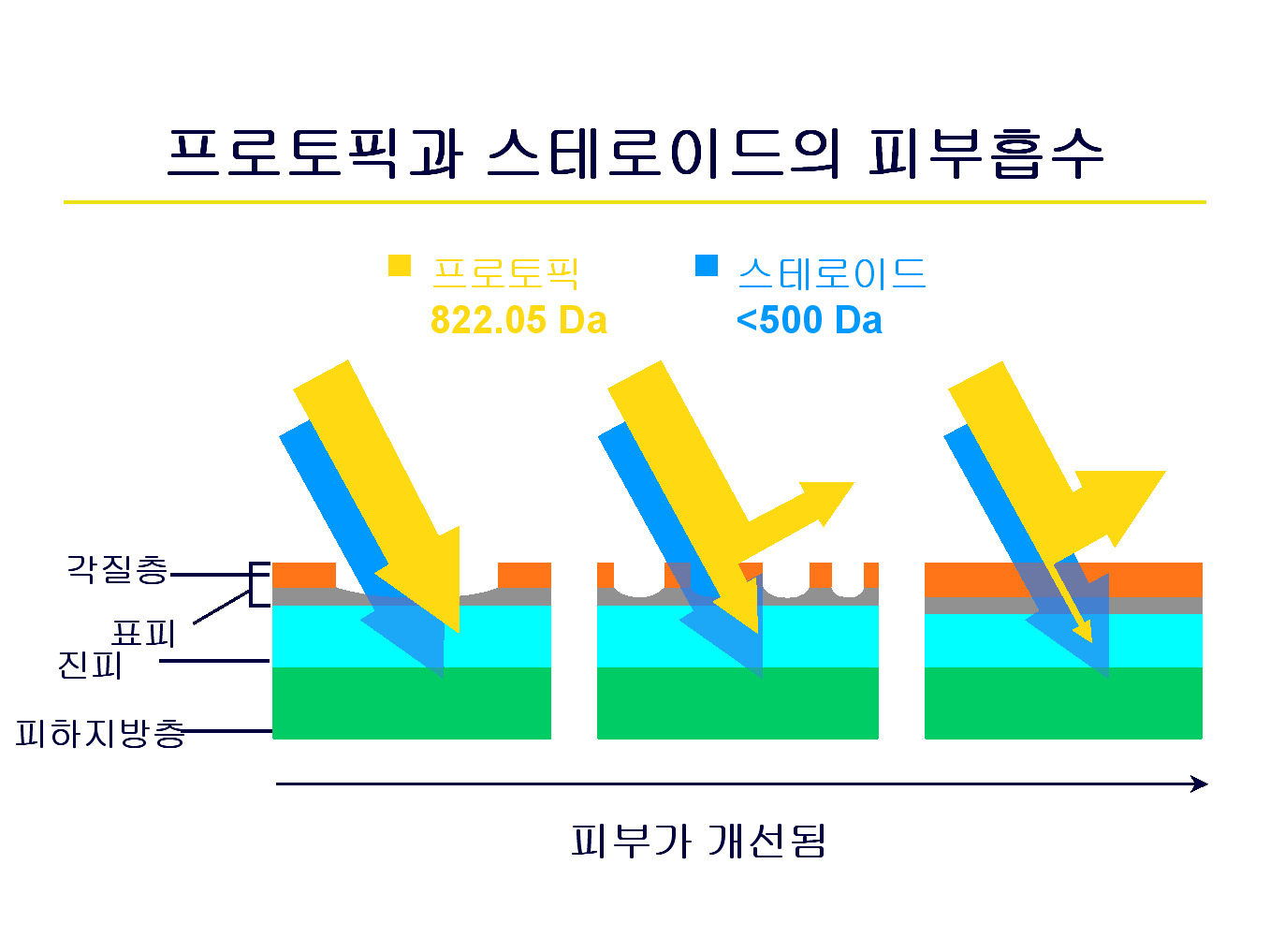
프로토픽은 분자량이 크고 피부 선택적(skin selective)이어서 아토피피부염 병변이 있는 곳에서 흡수가 이루어지며,
피부가 개선이 되어 건강해지면 흡수가 되지 않는 반면, 스테로이드는 건강한 피부에도 흡수가 됩니다.
(임상2) 장기 전신 흡수 관찰 시험2)3)4)
대상: 성인(631명) 및 소아(351명) 아토피피부염 환자
시행약물: 타크로리무스 0.1% 및 0.03%
방법: 1일 2회 12주간 도포
결과: 소아는 100%, 성인은 99.7%가 혈중농도 5ng/ml미만
(임상3) 소아 전신 흡수 관찰 시험5)
대상: 2-15세 소아 아토피피부염 환자. 189명
시행약물: 타크로리무스 0.03%
방법: 1일 2회 3주간 도포
결과: 전체 환자의 98.4%가 혈중농도 1ng/ml 이하임.
가장 높게 측정된 레벨은 2.75ng/ml였음.
전신흡수를 나타나는 최소값은 5ng/ml으로 그 미만일 경우 면역억제를 하지 않는다고 볼 수 있습니다.
결론: 프로토픽은 아토피피부염의 병변에만 선택적으로 작용하여 최소의 전신흡수를 나타내어, 전신면역억제를 하지 않습니다.
발췌:
1) I. Berkersky et al, Nonclinical and early clinical development of tacrolimus ointment for the treatment of atopic dermatitis. J Am Acad Dermatol 2001; 44: S17-27
2) Jon M. Hanifin,MD. Tacrolimus ointment for the treatment of atopic dermatitis in adult patients: Part1, Efficacy, J Am Acad Dermatol,2001; 44: S28-38
3) Jon M. Hanifin,MD. Tacrolimus ointment for the treatment of atopic dermatitis in adult patients: Part2, Safety, J Am Acad Dermatol,2001; 44: S39-46
4) A 12 weeks study of tacrolimus ointment for the atopic dermatitis in pediatric patients. J Am Acad Dermatol,2001; 44: S47-57
5) T. Ruzicka, for the European/Canadian Tacrolimus ointment study group. Pediatric patients with moderate to severe atopic dermatitis treated with tacrolimus ointment 0.03% show minimal systemic exposure: poster from 20th WCD
|
|